
Western blot (WB) was used to confirm all samples with one or both EIA reactive or grey zone results and all samples with reactive Abbott CMIA results. Methods: HIV Ag/Ab chemiluminescence microparticle immunoassay (ARCHITECT CMIA, Abbott Diagnostics, a version of CLIA) was performed on 2,138 samples. 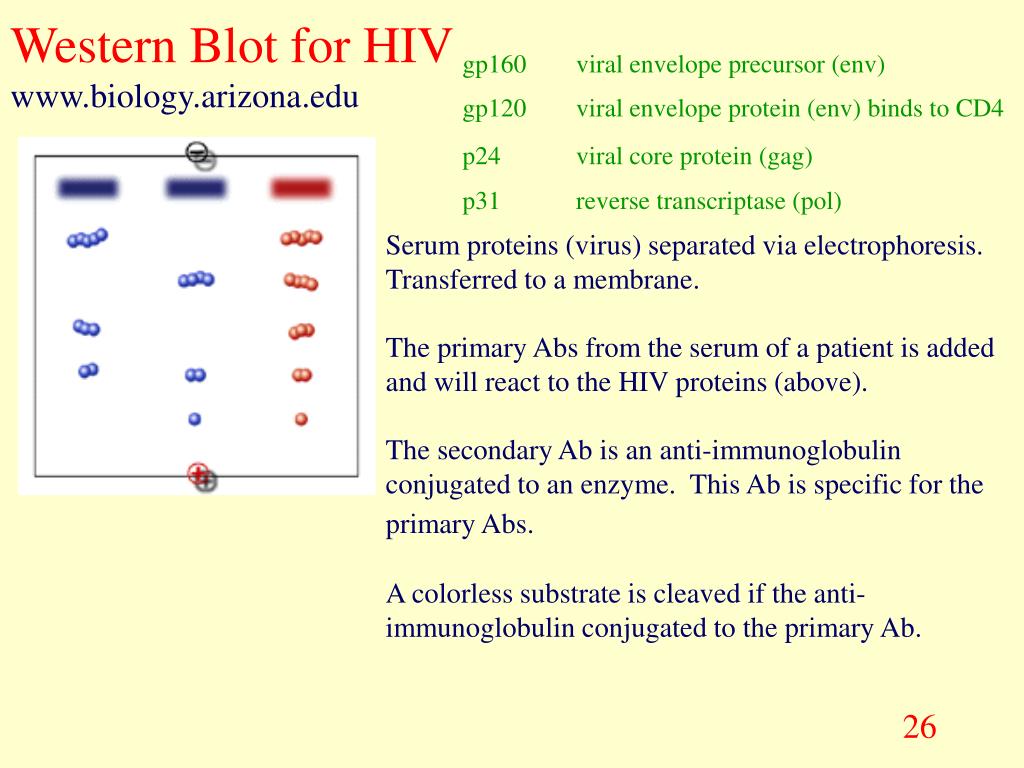
The aim of this study is to evaluate the performance of the three strategies. Currently, there is no public data to compare the performance of the two-different-EIAs strategy, the single-EIA and single-CLIA strategies for HIV donor screening in China. Mei-Yu Wang 1,2, Sushil Devare 3, Jian-Fang Liu 4, Xiao-Ting Lv 4, Peng Yin 4, Nan Guo 5, Ping Fu 1,2, Bin-Ting Wu 1,2, Yong-Hua Yin 1,2, Ling Ke 1,2, Xue Li 1, Hua Shan 5, Yu Liu 1,2ġ Institute of Blood Transfusion, Chinese Academy of Medical Sciences & Peking Union Medical College, Chengdu 610052, China Ģ Sichuan Blood Safety and Blood Substitute International Science and Technology Cooperation Base, Chengdu 610052, China Ĥ Abbott Diagnostics, Shanghai R&D Center, Shanghai 200003, China ĥ Transfusion Medicine Service, Stanford University Medical Center, California, USAĬontributions: (I) Conception and design: H Shan, S Devare, Y Liu, P Yin (II) Administrative support: JF Liu, XT Lv, P Yin, H Shan (III) Provision of study materials or patients: Y Liu (IV) Collection and assembly of data: MY Wang, Y Liu (V) Data analysis and interpretation: MY Wang, Y Liu (VI) Manuscript writing: All authors (VII) Final approval of manuscript: All authors.īackground: Preventing transfusion-transmitted HIV infection requires a sensitive and specific donor screening strategy. Policy of Dealing with Allegations of Research Misconduct.Policy of Screening for Plagiarism Process.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
